“Lies, damned lies, and statistics.”
CDC’s intermediate term (minimum 90 days ) follow-up and outcomes of myocarditis reports in VAERS: far worse than previous claims of “generally mild”
INTRODUCTION
We have heard the incessant, perfunctory refrains “safe and effective” and “generally mild” so often, one wonders if these are preprogrammed slogans for Public Health rhetoric regardless of actual published data on vaccine associated myocarditis (VAM). CDC published finalized analysis of VAERS data on myocarditis after COVID-19 vaccination in Lancet this week. ACIP presentations have given us glimpse of this data over the past year; however, this formal analysis truly underscores CDC’s biased selective analysis to advance an oversimplified agenda that may be causing irreparable harm. Any glimmer of hope that the agency was aiming for redemption by acknowledging the very public missteps made throughout the pandemic are stultified by their ongoing dismissiveness toward the severe adverse reactions to COVID-19 vaccinations, especially myocarditis in children and young adults. The latest CDC publication is a thinly vailed attempt at having nothing more than a pretense of rigorous pharmacovigilance to ensure public safety.
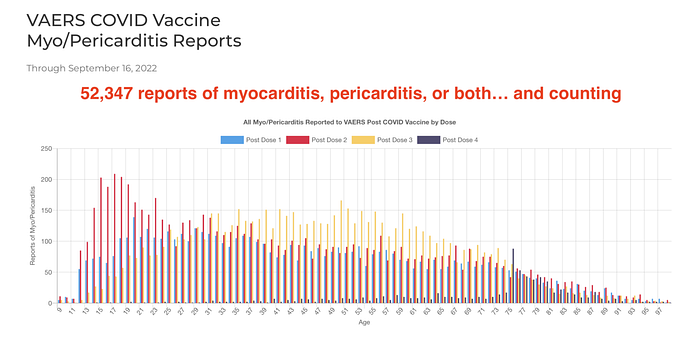
In reviewing the study, one must be mindful that data from Vaccine Adverse Event Reporting Syste (VAERS) alone under estimate the risk of VAM by three to four times, as I have reviewed in great detail previously (here and here). Furthermore the severity of VAM in VAERS is probably underestimated, as the gravity of the condition seems much greater than the oft repeated “generally mild” by CDC. For example, preliminary VAERS data (as reported in ACIP presentations) reveals that 2% of VAM patients required admission into the Intensive Care Unit (ICU). However, data from Vaccine Safety Datalink (VSD) demonstrates that VAM after second dose resulted in 34% requiring ICU admission. After the booster (third) dose a staggering 50% required ICU admission. Also, CDC’s analysis of VAERS data excludes those who may have died prior to hospitalization (and hence the diagnosis of ‘myocarditis’ might have been missed). Finally, while CDC continues to recommend COVID-19 vaccination for everyone, regardless of risk stratification by age and sex, recent research from UK and this preprint analysis on risks and benefits by age and sex indicate there may be a net harm in children and young adults compared to COVID-19 infection itself.
STUDY DESIGN AND METHODOLOGY

In this follow-up surveillance study, CDC conducted surveys in US individuals aged 12–29 years with myocarditis after mRNA COVID-19 vaccination (from reports in VAERS between Jan 12 and Nov 5, 2021). VAERS is a national passive surveillance system coadministered by the CDC and the US Food and Drug Administration (FDA). A two-component survey was administered: one component to patients (or parents or guardians) and one component to health-care providers, to assess patient outcomes at least 90 days since myocarditis onset. Data collected were recovery status, cardiac testing,functional status, and EuroQol health-related quality-of-life measures, and a weighted quality-of-life measure. The EuroQol results were compared with published results in US populations (aged 18–24 years) from before and early on in the COVID-19 pandemic. Physicians at the CDC reviewed all identified VAERS reports and available medical records to determine if the case met CDC case definition criteria for confirmed or probable myocarditis or myopericarditis (henceforth referred to as myocarditis.
Ignoring the preponderance of evidence from Hong Kong, Israel, UK, Ontario (Canada), four Nordic countries, USA, and even their own CDC MMWR data from 40 health care system databases, in this analysis of VAERS data, CDC once again proclaims, “Despite the higher than expected occurrence of myocarditis after COVID-19 vaccination, the benefits of mRNA COVID-19 vaccines have been shown to outweigh the risk of myocarditis.” An outstanding detailed and quantified analysis of the risk and benefits of COVID-19 vaccination in children and young adults is in stark contrast to CDC’s erroneous conclusion (which was based upon selective data analysis with severe limitations). To date, neither CDC nor any organization mandating COVID-19 vaccination (e.g., state public health departments, employers, schools and universities, and competitive athletic events) have ever published such a comprehensive analysis to justify their claims and mandates. To be clear, for those who are 65+ years old and those who have underlying medical conditions, the benefits (reduction in hospitalization and death from COVID-19 infection) of COVID1–19 vaccination probably do outweigh the risks. However, for healthy children and young adults there seems to be strong support to indicate a net harm (and as this follow-up study reveals, that harm includes long term heart damage).
From Aug 24, 2021, to Jan 12, 2022, CDC administered a two-component telephone survey to assess patient outcomes at least 90 days since the onset of myocarditis symptoms after mRNA COVID-19 vaccination . The first component administered to adult patients or to the parents or guardians of minor patients ascertained quality of life, previous medical history, need for ongoing medication for myocarditis, and presence of clinical symptoms in the 2 weeks before the date of the survey, including chest pain, shortness of breath, fatigue, and palpitations, hospitalisations, and days of school or work missed in the 2 weeks before the survey. The second component of the two-part survey was a survey of health-care providers who provided care to eligible patients for this study with myocarditis after mRNA COVID-19 vaccination, which ascertained patient cardiac health and functional status. Follow-up assessments of cardiac health after the initial myocarditis diagnosis or hospitalisation for myocarditis after COVID-19 vaccination included findings from electrocardiograms, echocardiograms, cardiac MRIs, troponin concentrations, exercise stress testing, and ambulatory rhythm monitoring. Assessments of functional status included ongoing treatment or health-care provider-recommended restrictions on physical activity.
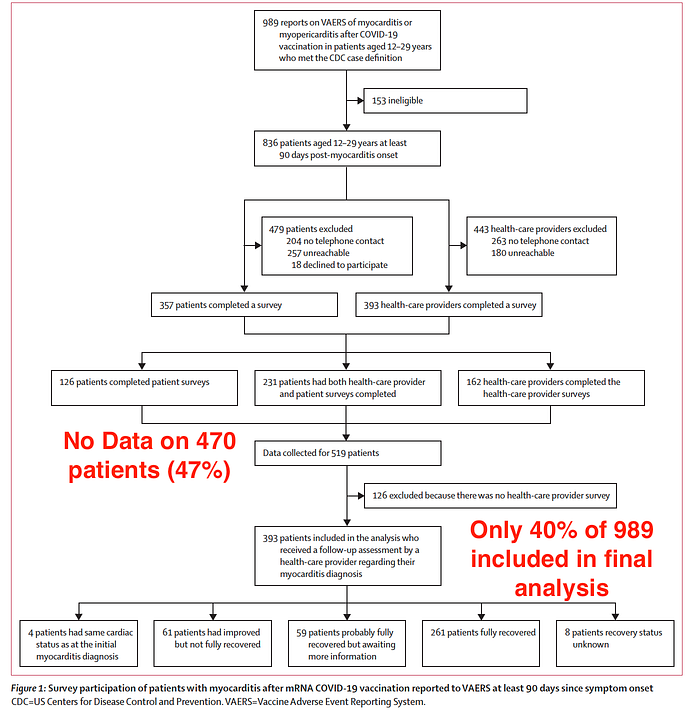
Figure 1 summarizes the inclusion and exclusion criteria. Glaringly and disturbingly obvious is that no data are available for 47% of the patients. These patients (or health care providers) either had no telephone number or were unreachable. Why? This is worrisome for professional neglect and lack of research integrity on the part of CDC. Why were 257 patients not reachable? Is it possible they died? Did CDC verity with social security death index? Did CDC send local law enforcement of social workers for a ‘wellness check’ to ensure patients were still alive? Outcome analysis might be severely altered (possibly demonstrate greater severity of myocarditis) if CDC had follow-up data on these 470 patients who were ‘missing’ telephone contact information or were ‘unreachable’. Does CDC just not want the public to know?

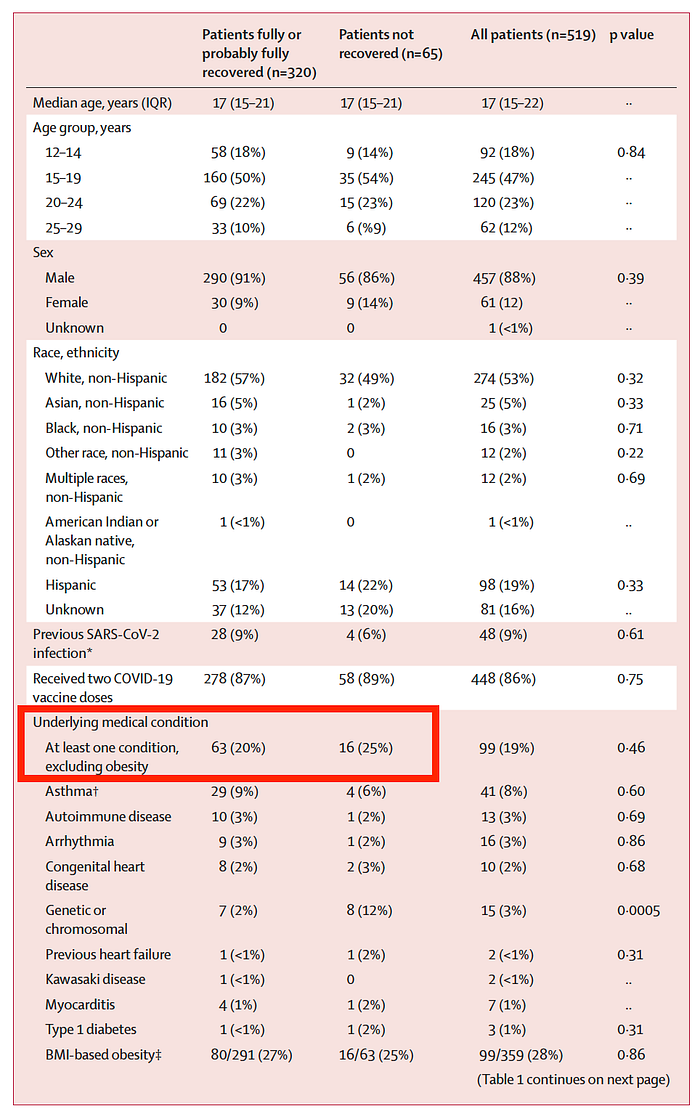
Also, noteworthy is that only 20% had underlying medical conditions (additional 27% had obesity). By comparison, 92% of all those hospitalized for COVID-19 infection had at least one underlying medical condition (and 50% of all COVID-19 hospitalizations were those over 65 years old). As many of us had admonished: myocarditis after COVID-19 vaccination tends to occur in younger healthier individuals. Severe COVID-19 complications tend to occur in older people (or younger people with underlying medical conditions). Therefore, vaccine mandates for children and young adults are likely causing disproportionate unethical harm in those who are at very low risk of severe COVID complications themselves (more on children and COVID-19 here).
FINDINGS AND RESULTS
As summarized in Figure 1 above: Between Jan 12 and Nov 5, 2021, 989 cases of myocarditis after mRNA COVID-19 vaccination in patients aged 12–29 years were reported to VAERS and met the CDC’s case definition for confirmed or probable myocarditis. Of these, 836 (85%) patients were at least 90 days post-myocarditis onset (figure 1). Median interval from myocarditis onset to survey completion was 143 days for patients and 191 days for health-care providers. Patients with a left ventricular ejection fraction (LVEF) of less than 50% were analyzed. Of the 100 survey respondents with LVEF values recorded at their initial diagnosis, 33 (33%) had LVEF values less than 50%, which was not statistically different from the results in non-respondents (27 [42%] of 65 non-respondents. While not statistically significant, 42% versus 33% is worrisome (i.e., a greater percent of non-respondents had low LVEF). Low LVEF can be a risk factor for cardiac arrest and sudden cardiac death (SCD) and therefore this should have been further discussed and investigated.
For 393 (47%) of 836 patients, health-care providers were contacted; 241 (61%) of 393 were cardiologists. At the time of the survey, health-care providers considered 320 (81%) of 393 patients to be recovered: 261 (66%) patients were considered fully recovered and 59 (15%) patients were considered to be probably recovered but awaiting further information. However, at the last health-care provider follow-up, 104 (26%) of 393 patients were prescribed daily medication related to myocarditis. An additional 61 (16%) patients had improved and four patients had the same cardiac status as at the initial myocarditis diagnosis; these 65 patients were categorised as not fully recovered (figure 1).
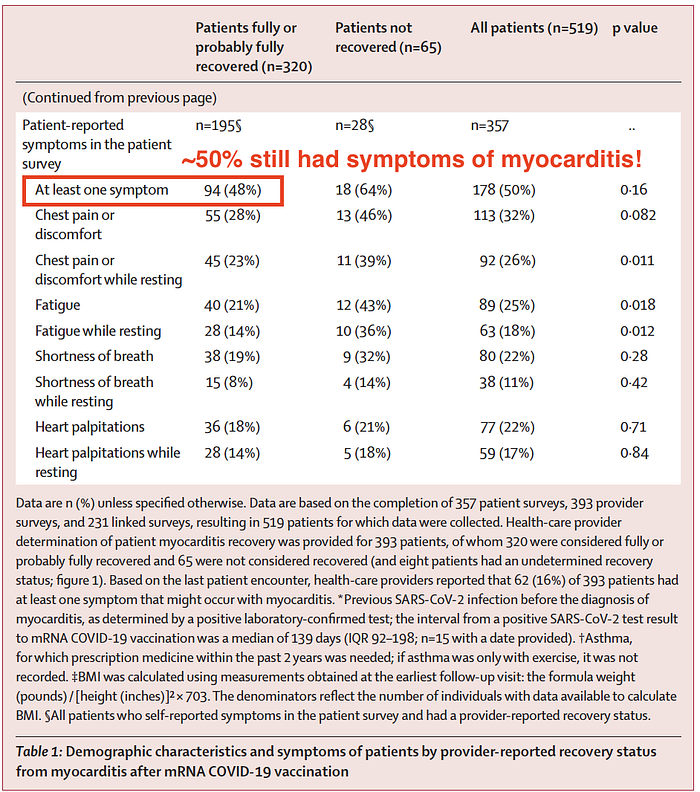
In the 2 weeks before the survey date, 178 (50%) of 357 patients reported having at least one symptom that might occur with myocarditis (chest pain or discomfort, fatigue, shortness of breath, or palpitations). Patients who were not considered recovered from myocarditis more frequently reported fatigue than did patients who were considered recovered (12 [43%] vs 40 [21%]; p=0·018; table 1). By contrast, based on the last patient encounter, health-care providers reported that 62 (16%) of 393 patients at least one symptom that might occur with myocarditis (table 1).
Of 357 patients surveyed, 267 (75%) were enrolled in school or in paid employment; 43 (16%) of whom reported missing school or workdays in the 2 weeks before the survey date. Of those 43 patients, 15 (35%) believed it was associated with myocarditis.
Most patients were admitted to hospital after an initial diagnosis of myocarditis (484 [93%] of 519 patients). Of these 484 patients, 393 (81%) patients had information on level of care, according to the health-care provider surveys; 99 (25%) of these 393 patients were treated in an intensive care unit. [Note: in all the ACIP presentations, CDC prelim analysis indicated <2% required ICU care.] To our knowledge, no deaths occurred during follow-up among the patients eligible for the survey. Notably, CDC does not seem to consider that at least some of those lost to follow-up could have died. Not mentioned in this study by CDC is that National Center for Health Statistics (NCHS) apparently notes 67 death certificates with COVID-19 vaccination mentioned as cause or as a co-morbid condition. Also, recent study from UK published in Circulation noted 100 deaths after myocarditis from COVID-19 vaccination (up to 28 days after hospitalization). Prior research has noted that the risk of sudden cardiac death persists for up to 6 months after diagnosis of myocarditis leading to activity restriction recommendations. CDC’s approach to verifying deaths after myocarditis from COVID-19 vaccination (lack of concerted effort via comprehensive and methodical investigation) is disingenuous at best and unethically incompetent at worst.

Six (2%) of 357 patients who self-reported re-admission to hospital had a hospital admission because of an adverse event after myocarditis treatment (n=3; adverse reactions to intravenous immune globulin) or had any cardiac abnormality identified (n=3; appendix 1 p 6); all patients were discharged within 1 week.
At follow-up, fewer patients had restrictions on physical activity than at initial diagnosis, and 34 (52%) of 65 individuals with restrictions on physical activity at the time of follow-up who were not considered recovered were cleared for all physical activity; 31 (48%) individuals still had restrictions (table 2). 91 (28%) of those considered fully or probably fully recovered continued to have activity restrictions at follow-up. Again, 48% of those not fully recovered and 28% of those fully or probably fully recovered continued to have activity restrictions at median follow-up of 98 days!!!

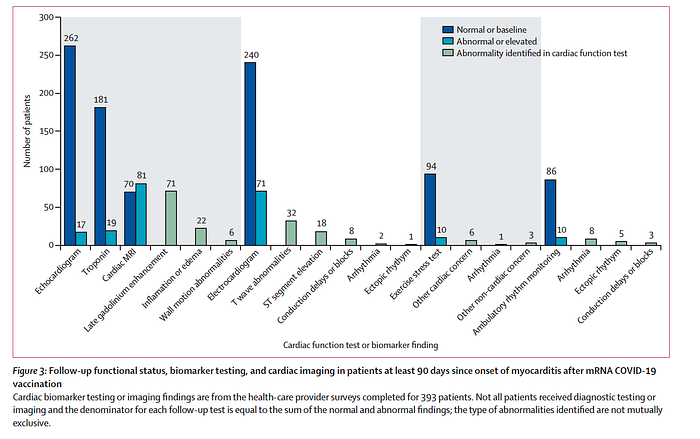
Among the 151 patients who had cardiac MRIs (CMR) during outpatient follow-up, 81 (54%) patients had one or more abnormalities. Abnormal cardiac MRI findings included the presence of late gadolinium enhancement (71 [47%] patients), inflammation or edema (22 [15%] patients), or wall motion abnormalities (six [4%] patients). Evidence of ongoing myocarditis, defined by both late gadolinium enhancement and edema using modified Lake Louise criteria, occurred in 20 [13%] of 151 patients. Previous research has demonstrated persistent abnormal CMR findings in about 75% of adolescents 3–8 months after initial diagnosis of VAM.
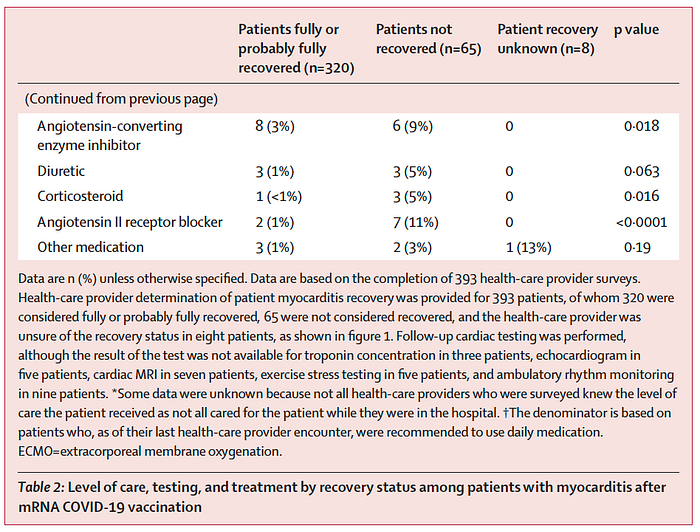
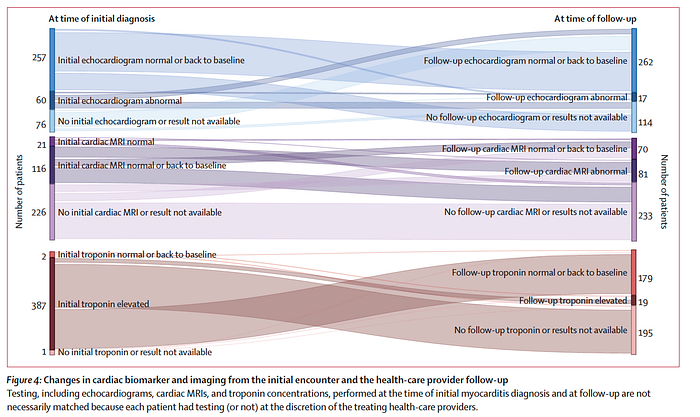
In the subset of patients with abnormal findings at the time of myocarditis diagnosis, abnormal diagnostic markers or abnormal imaging were also observed at follow-up in seven (12%) of 60 with initial abnormal echocardiograms, 19 (5%) of 387 with initial elevated troponin levels, and 47 (32%) of 146 with initial abnormal cardiac MRIs (figure 4). All cardiac test results (ie, echocardiogram, electrocardiogram, cardiac MRI, and troponin) were available for follow-up review in only 199 (62%) of 320 patients considered recovered, 44 (68%) of 65 considered not recovered, and three (38%) of eight with an unknown recovery status. In other words, complete diagnostic testing was not performed or not available in 38% of those considered recovered and 32% of those considered not recovered.
SUMMARY AND KEY POINTS
Since April 2021, when early reports of vaccine associated myocarditis from Israel surfaced CDC has continued to be dismissive of both the incidence and severity. This CDC study is grossly flawed in its methodology. 60% of reported cases of myocarditis were not even included in the final analysis. Those who were not reachable may have died (needs to be more thoroughly investigated). Many (38%) did not have complete diagnostic data at time of follow-up. Even with the limitations noted above:
- 99 (25%) of these 393 patients were treated in an intensive care unit
- A staggering 50% still had ongoing symptoms of myocarditis
- 48% of those not fully recovered and 28% of those fully or probably fully recovered continued to have activity restrictions at median follow-up of 98 days
To continue to recommend COVID-19 vaccination (and boosters) for everyone, regardless of risk stratification by age and sex, and to continue the incessant perfunctory refrains “safe and effective” and “generally mild” is nothing short of egregious affront on medical ethics, thereby further eroding trust in Public Health institutions.
